Pollen movement in flowering plants depends on various vectors including animals, wind, and water. Compared to wind- or water-pollinated flowers, animal-pollinated flowers are generally showier, often with bright colors. They also often produce nectar or other rewards to attract pollinators. However, the advertisement and reward for pollinators may also attract plant enemies. Herbivores can consume parts of flower structures, entire flowers, or whole plants. For example, nectar robbers may penetrate a hole in the corolla and thereby suck nectar from flowers without playing a pollination role.
The great diversity found in angiosperm flowers can be fully understood only when the diverse floral traits are considered as functional units, shaped by partly opposing selective pressures (Faegri and van der Pijl 1979; Waser and Ollerton 2006). In experimental work on the evolution of floral traits, most attention has centered on natural selection that favors mutualistic pollinators and hinders antagonistic herbivores (Fenster et al. 2004; Strauss and Whittall 2006).
A Closer Look at Dove Tree
The dove tree, Davidia involucrata, is a species prized by gardeners for its showy bracts. The only species in the genus, dove tree is a medium-sized tree (up to 20 meters [65 feet] tall) that is endemic in mountain forests at altitudes of 1,100 to 2,600 meters (3,600 to 8,500 feet) in western China (Fang and Chang 1983). Fossils from the Paleocene of North America indicate that the lineage was more widespread in the past (for a review, see Manchester 2003).
The genus Davidia is named after Father Armand David (1826–1900), a French missionary and keen naturalist who lived in China from 1862 to 1874 and collected many specimens of plants previously unknown in the West (Zhang and Li 1994). David was also the first westerner to describe another rare Chinese endemic, the giant panda (Ailuropoda melanoleuca).


The dove tree has been considered a first-class endangered plant in China, but it became well-known in its homeland only after it was seen growing in other countries during two diplomatic visits by Chinese leaders. When Enlai Zhou (1898–1976), the first premier of the People’s Republic of China, visited Geneva, Switzerland, in 1954, he was impressed by the beauty of flowering dove trees in the gardens of many local families and was told that the tree came from China. Another surprise happened when Chinese leaders visiting Washington, D.C., saw dove trees in bloom in front of the White House in the early 1970s.
The tree’s common names—dove tree or handkerchief tree—refer to the two white, paperlike bracts that surround the base of each flower head (capitulum). The bracts initially are small and green, resembling leaves, but increase in size and turn white as the flowers mature. The change in bract color from green to white is associated with the bracts becoming UV (ultraviolet)-light-absorbing (Burr and Barthlott 1993). The anthers are even more strongly UV-light-absorbing. This trait is associated with attracting pollinating insects that see UV light, and the species has therefore been classified as entomophilous (insect pollinated) (Burr and Barthlott 1993), although the pollination of Davidia involucrata had not been previously studied in the field. We are particularly interested in the questions “What is the function of the white bracts? Do the dove tree’s bracts play a role in attracting pollinators?”
Many plants within Cornales, the dogwood clade, have large bracts that surround the inflorescences. Bracts have been thought to protect flowers from herbivores in various species. For example, the bracts of Dalechampia vines function as honest signals to pollinators of the presence of floral reward (Armbruster et al. 2005) and also function in protection from florivores (flower eaters) and pollen thieves (Armbruster 1997).

Flowers are also under strong selection pressures from their physical environment, yet research on selection by abiotic environmental factors on flowers has been limited (Corbet 1990; Galen 2005). During our previous field work, our observation was often interrupted by rainy days. Rain is one of those abiotic factors that acts as a selective agent on flowers. This was first recognized by Sprengel ([1793]1972), who noted that rain may wash away pollen grains and dilute flower nectar. This added another question to our research: Did the need to protect pollen grains from rain play a role in the evolution of large bracts?
Carrying Out the Study
With our questions about rain protection and pollinator attraction in mind, we carried out observations and experiments in a natural population of dove trees in western China (Shennongjia Nature Reserve, Hubei) in April 2005 and April 2006. In our study, we manipulated flowers in a population of 16 flowering dove trees to assess the function of this species’ bracts.
Though dove tree’s mature bracts draw much attention, the round inflorescences dangling beneath the bracts are also interesting. The dark purple inflorescences generally consist of a single perfect (holding both stamens and pistils) flower surrounded by numerous male flowers (Fang and Chang 1983). The individual flowers are naked and nectarless, without sepals or petals. Before flowering, dove tree’s bracts are green, turning white rapidly as flowers mature and anthers begin to dehisce. The anthers split longitudinally, and pollen grains are exposed on the recurved anther walls. Individual capitula of the dove tree last 5 to 7 days and the bracts drop off when flowering ends.
As part of the study, we collected both green and white bracts and preserved them for later observation under the microscope. We also measured the length and width of the bracts on each of 20 capitula daily between April 16 and April 22, 2006.
Do Dove Tree’s Bracts Function in the Pollination Process?
To investigate the role of bracts in pollinator attraction, we recorded pollinator visits to four kinds of capitula: (1) natural, (2) with both bracts removed, (3) with both bracts replaced by green artificial bracts made of copy paper, or (4) with both bracts replaced by white artificial bracts made of copy paper. The artificial bracts were similar in shape and size to the natural bracts. We recorded the number of pollinator visits to the dove trees’ flowers from 10:00 in the morning to 3:00 in the afternoon between April 18 and April 22 in 2005, and between April 16 and April 20 in 2006. Two observers monitored two sites during these periods of maximum pollinator activity. Insect visits became extremely rare after 3:00 in the afternoon, and during one night of observations, no pollinators were observed visiting this nectarless species. Pollinators were collected and sent to the Institute of Zoology of the Chinese Academy of Science in Beijing for identification.

To examine whether capitula with intact bracts lost more or less pollen to rain than did capitula that had their bracts removed, late in the flowering period we counted the pollen remaining within 29 and 27 capitula that were natural or had both bracts removed, respectively, and had experienced at least one rainy day.
Numbers of anthers and ovules per capitulum and pollen grains produced per anther were estimated in 34 capitula with freshly dehiscing anthers. In September 2005 and 2006, we collected 39 and 32 open-pollinated capitula and counted their seeds to estimate seed set under natural pollination.
Pollen Stickiness and Behavior in Water
The pollen grains of anemophilous (wind pollinated) plants are not sticky, while those of entomophilous plants usually are sticky in order to adhere to visiting pollinators. To assess the possibility of wind pollination of dove tree, we tested the stickiness of its pollen grains by their adherence to glass slides. We also placed netting around 20 capitula, thereby excluding pollinators but allowing possible wind pollination, and later examined the netted capitula for seed production.
To test the behavior of pollen grains under rainy conditions, we followed Huang et al.’s (2002) method of pollen germination. Pollen grains from newly dehisced anthers were placed in sucrose solutions of 5%, 10%, 15%, and 20% by mass to examine optimum conditions for pollen germination. We then compared germination rates of pollen grains from eight randomly collected capitula in distilled water or in the optimum sucrose solution (10%). Pollen grains that had either germinated or burst after 4 hours were counted under a light microscope. Ten samples of pollen grains from each flower were analyzed in this manner. Data analysis was accomplished by using one-way ANOVA analysis to compare the frequencies of pollinator visits to the four kinds of capitula and the amount of pollen remaining in capitula after different experimental treatments and to assess pollen viability in distilled water versus 10% sucrose solution.
What We Found Out…
When we looked at the dove tree bracts that we had collected at either the green or white stage, we saw that the parenchyma cells in the green bracts were full of chloroplasts, while the parenchyma cells of white bracts had degenerated and contained few chloroplasts. Bract color turned from green to white on the third or fourth day, when the anthers began to dehisce and when bracts had reached three-quarters of their final size. Bracts continued to grow during anthesis but dropped off soon thereafter.
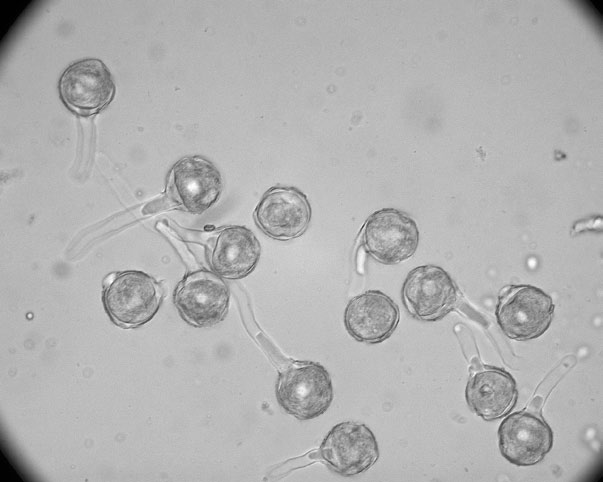
Pollen-collecting bees and pollen-feeding beetles were the major visitors and pollinators of the flowers. We recorded a total of 2,174 visits to capitula, of which bees and beetles accounted for 93.6% and 6.4%, respectively. Beetles generally stayed within a capitulum for 1 to 2 hours, while bees spent 4 to 6 seconds per capitulum foraging for pollen, suggesting that bees are the more effective pollinators of the dove tree. Bees that visited included Apis cerana, Xylocopa appendiculata (Apidae), and Halictus and Lasioglossum species (Halictidae), and beetles included Agriotes species (Elateridae), Oxycetonia jucunda (Cetoniidae), and species of Nitidulidae. Visitor frequencies were low, and so was seed set of open-pollinated capitula in both years. It seems clear that wind pollination of dove tree is unlikely since its pollen grains are sticky and apparently not picked up by wind. Also, the netted capitula did not produce any seeds, further evidence that insects rather than wind pollinate dove tree flowers.
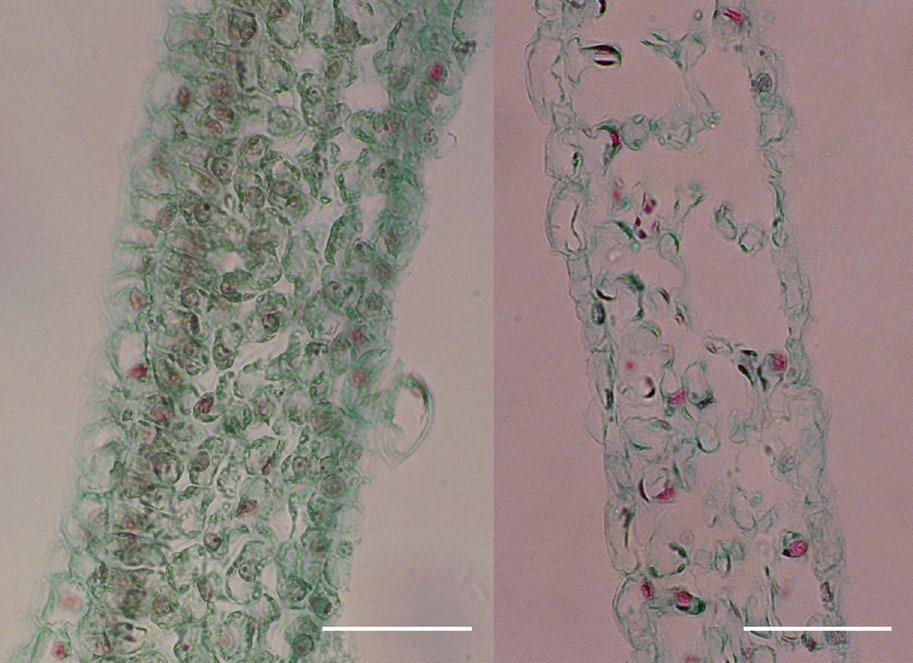
Bees preferred to visit capitula with white bracts over those with green bracts, on the basis of four consecutive days of observations on inflorescences of 62 natural capitula, 62 bractless capitula, 62 white-papered capitula, and 48 green-papered capitula. Visits to natural and white-papered inflorescences were not significantly different but were higher than those in the other two treatments. Visits to inflorescences with bracts removed and those with bracts replaced by green paper did not differ significantly.

The pollen/ovule ratio in D. involucrata is extremely high, with a capitulum producing only about 7 ovules to about 900 to 1,000 anthers, which produced over a million pollen grains in total. Pollen amounts remaining in capitula with their bracts removed were not significantly different from those in intact capitula but were significantly lower than the total pollen production, demonstrating high pollen loss to rain or removal by pollinators.
Pollen germinated best in 10% sucrose solution, and there was a highly significant difference in germination and bursting rates between pollen grains placed in distilled water or 10% sucrose solution. After 4 hours, about 85% of pollen grains in water had burst, while only about 37% of the grains in 10% sucrose solution had burst. (ed. note: see Sun et al. 2008 for detailed results).
… and What It Means
Pollen-collecting bees, the most important pollinators of Davidia involucrata, preferred visiting white-bracted capitula, and it is therefore likely that during their green stage the bracts function in photosynthesis, while during their white, UV-light-absorbing stage their function changes to attracting pollinators. During more than 170 hours of observation, we never observed bees visiting green-bracted capitula. Their UV-light absorbance makes the white bracts stand out from surrounding foliage in the bee visual spectrum (Burr and Barthlott 1993; Kevan et al. 1996) and is likely due to the flavonoids that are the major pigments in the bracts of D. involucrata (Hu et al. 2007).

Over the 2 years of our study, insect visitation was low. With low visitation rates, prolonging the flowering period will benefit reproductive success as a sit-and-wait strategy (Ashman and Schoen 1994). Protection of the pollen grains presented on the recurved anther walls during the 5 to 7 day flowering period would then be of key importance; the longer viable pollen is present, the better the chance of a pollinator visiting within that period. Since dove tree’s flowers lack a corolla, protection of the pollen must be achieved by the bracts. Dove trees generally flower from mid-April to mid-May, a period which is within the rainy season of the subtropical region where they occur. Since the pollen grains of dove tree readily burst in water, it is probable that the rooflike bracts function as an umbrella to reduce rain damage to the anthers and pollen. Indeed, we repeatedly observed rain-damaged stamens in the capitula where we had removed bracts, while capitula with intact bracts had dry stamens even after heavy rain. The fact that bract-bearing capitula lost as much pollen as bractless ones is attributable to bees removing most pollen from the former, while rain washed away most pollen from the latter.
The evolution of flower or inflorescence structures with multiple functions may reflect the net effect of conflicting or additive selective pressures (Anderson 1976; Armbruster 1996, 1997, 2001; Galen 1999; Fenster et al. 2004; Armbruster et al. 2005; Strauss and Whittall 2006; Waser and Ollerton 2006). In the case of the dove tree, pollinator selection favors white bracts, while the need to protect pollen from rain favors large bracts. Notably, dove tree’s two bracts differ in size, perhaps in response to constraints on weight or resources.
Experimental studies of the effects of rain on floral traits are scarce (Bynum and Smith 2001; Huang et al. 2002; Galen 2005). Recent studies of pollen longevity in 80 angiosperms found that pollen life-span was decreased by direct contact with water (Mao and Huang 2009). Another evolutionary tactic is seen in the erect flowers of Primula vulgaris; though the flowers regularly fill with water, roughly a quarter of the primula’s pollen grains will still germinate even after 5 hours underwater (Eisikowitch and Woodell 1975). While some species have relatively high water-resistant pollen, others rely on nodding flowers, or close their flowers by petal movements (Bynum and Smith 2001; Hase et al. 2006; He et al. 2006), or have other features protecting pollen from wetting, such as in the dove tree.

It seems that the bracts of the dove tree are a striking example of the multiple roles played by one structure in photosynthesis, pollinator attraction, and as a stamen rain shelter. As another example, in Tilia the bracts not only help wind dispersal of the fruits once they mature, but also may act as flags that attract pollinators—especially nocturnal pollinators—to the flowers (Anderson 1976). Our observation of the multifunctional nature of bracts in the dove tree suggested that floral traits are under selection pressure from mutualists and antagonists as well as selection by abiotic environmental factors.
Acknowledgments
Citation: Sun, J. F. and Huang, S. Q. 2011. White bracts of the dove tree (Davidia involucrata): Umbrella and pollinator lure? Arnoldia, 68(3): 2–10.
This public introduction of our work in the dove tree is based on the first author’s PhD thesis and a paper in American Naturalist (2008). We thank Y.-B. Gong, Q. Fang, and S. S. Renner for collaboration, and we also thank L. Wang from the Altar Park in the Shennongjia Nature Reserve as well as W. Chen, Q. Li, Y.-Y. Mao, and M.-Q. Pan from Wuhan University for help in the field and in the lab, and S. Armbruster and an anonymous reviewer for many valuable suggestions for an early draft. A grant from the National Science Foundation of China (30825005) to S.-Q. Huang supported this work.
Literature Cited
Anderson, G. J. 1976. The pollination biology of Tilia. American Journal of Botany 63: 1203–1212.
Armbruster, W. S. 1996. Evolution of floral morphology and function: an integrative approach to adaptation, constraint, and compromise in Dalechampia (Euphorbiaceae). In: D. G. Lloyd and S. C. H. Barrett, eds. Floral Biology, pp. 241–272. Chapman & Hall, New York.
Armbruster, W. S. 1997. Exaptations link the evolution of plant-herbivore and plant-pollinator interactions: a phylogenetic inquiry. Ecology 78: 1661–1674.
Armbruster, W. S. 2001. Evolution of floral form: electrostatic forces, pollination, and adaptive compromise. New Phytologist 152: 181–183.
Armbruster, W. S., L. Antonsen, and C. Pélabon. 2005. Phenotypic selection on Dalechampia blossoms: honest signaling affects pollination success. Ecology 86: 3323–3333.
Ashman, T. L., and D. J. Schoen. 1994. How long should flowers live? Nature 371: 788–791.
Burr, B., and W. Barthlott. 1993. Untersuchungen zur Ultraviolettreflexion von Angiospermenblüten. II. Magnoliidae, Ranunculidae, Hamamelididae, Caryophyllidae, Rosidae. Trop. subtrop. Pflanzenwelt 87, Akad. Wiss. Lit. Mainz. F. Steiner Verlag, Stuttgart, 193 S.
Bynum, M.R., and W. K. Smith. 2001. Floral movements in response to thunderstorms improve reproductive effort in the alpine species Gentiana algida (Gentianaceae). American Journal of Botany 88: 1088–1095.
Corbet, S. A. 1990. Pollination and the weather. Israel Journal of Botany 39: 13–30.
Eisikowitch, D., and S. R. J. Woodell, 1975. The effect of water on pollen germination in two species of Primula. Evolution 28: 692–694.
Faegri, K., and L.van der Pijl. 1979. The principles of pollination ecology. 3rd ed. Pergamon Press, Oxford.
Fang, W.-P., and C.-Y. Chang. 1983. Flora Republicae Popularis Sinicae, vol. 52(2). Pages 157–159. Science Press, Beijing.
Fenster, C. B., W. S. Armbruster, P. Wilson, R. Dudash, and J. D. Thomson. 2004. Pollination syndromes and floral specialization. Annual Review of Ecology, Evolution and Systematics 35: 375–403.
Galen, C. 1999. Why do flowers vary? The functional ecology of variation in flower size and form within natural plant populations. Bioscience 49: 631–640.
Galen, C. 2005. It never rains but then it pours: the diverse effects of water on flower integrity and function. In: R. Edward and F. A. Bazzaz, eds. Reproductive allocation in plants, pp. 77–95. Elsevier Academic Press, Burlington, MA, USA
Hase, A. V., R. M. Cowling, and A. G. Ellis. 2006. Petal movement in cape wildflowers protects pollen from exposure to moisture. Plant Ecology 184: 5–87.
He, Y.-P., Y.-W. Duan, J.-Q. Liu, and W. K. Smith. 2006. Floral closure in response to temperature and pollination in Gentiana straminea Maxim. (Gentianaceae), an alpine perennial in the Qinghai-Tibetan Plateau. Plant Systematics and Evolution 256: 17–33.
Hu, J.-Y., S.-L. Zhang, Z.-X. Su, and Y.-M. Liao. 2007. Pollinator attraction by Davidia involucrata. I. Color. Journal of Plant Ecology 31: 166–171. (In Chinese with English Abstract)
Huang, S.-Q., Y. Takahashi, and A. Dafni. 2002. Why does the flower stalk of Pulsatilla cernua (Ranunculaceae) bend during anthesis? American Journal of Botany 89: 1599–1603.
Jones, C. E. 1967. Some evolutionary aspects of a water stress on flowering in the tropics. Turrialba 17: 188–190.
Kevan, P., M. Giurfa, and L. Chittka. 1996. Why are there so many and so few white flowers. Trends in Plant Science 1: 280–284.
Mao Y.-Y., and S.-Q. Huang. 2009. Pollen resistance to water in 80 angiosperm species: flower structures protect rain susceptible pollen. New Phytologist 183: 892–899.
Manchester, S. R. 2003. Leaves and fruits of Davidia (Cornales) from the Paleocene of North America. Systematic Botany 27: 368–382.
Sprengel, C. K. 1793. Das entdeckte Geheimnis der Natur im Bau und in der Befruchtung der Blumen (Reprinted 1972). Weldon & Wesley, New York.
Strauss, S. Y., and J. B. Whittall. 2006. Non-pollinator agents of selection on floral traits. In: L. D. Harder and S. C. H. Barrett, eds. Ecology and evolution of flowers, pp. 120–138. Oxford University Press, Oxford.
Sun, J.-F., Y.-B. Gong, S. S. Renner, and S.-Q. Huang. 2008. Multifunctional bracts in the dove tree Davidia involucrata (Nyssaceae: Cornales): rain protection and pollinator attraction. The American Naturalist 171: 119–124
Waser, N. M., and J. Ollerton. 2006. Plant-pollinator interactions: from specialization to generalization. University of Chicago Press, Chicago.
Zhang, J., J. Li, and X. Lian. 1994. Morphology and biological characteristics of Davidia involucrata. Journal of Beijing Forestry University 16: 33–37
Ji-Fan Sun and Shuang-Quan Huang are from the College of Life Sciences at Wuhan University in Wuhan, China.