Oaks afford a unique insight into the history of our landscape, flora, and vegetation. Oaks have been among the dominant trees of eastern North American forests and woodlands for approximately 10,000 years (Abrams 1992). Between 8,000 and 3,000 years ago, oaks spread to distributions close to those we observe today (Webb 1981). The landscape between the prairies of the Great Plains and the eastern deciduous forest had by that time settled into a broad transition zone in which prairie, woodland, and savanna shifted with the dynamics of climate and fire (Anderson and Bowles 1999). Some oak species in this region could persist below ground for decades as their shoots were regularly burned to the ground, growing to maturity only when a break in fire frequency allowed their stump sprouts to grow (Kline 1997). The oldest oaks still growing have borne witness to fires, changes in forest structure and composition, and substantial anthropogenic landscape changes. These old oaks sustain large numbers of mammals, birds, and insects. Blue jays, squirrels, and, previously, passenger pigeons have eaten, hoarded, and dispersed acorns in vast quantities (Johnson and Adkisson 1986; Keator and Bazell 1998; Price 1999, ch. 1), and civilization rests in part on the structural and nutritional properties of oaks (Logan 2005). It is hard for a North American naturalist to imagine a landscape without oaks.
At the same time, oaks are remarkable for their ability to stump botanists. Even where there are only a few species to choose from, we often struggle to put a name on oaks in the field, and annotations on many herbarium specimens capture decades of disagreement. Oaks are notoriously promiscuous, with closely related species able to exchange genes seemingly at will. Pioneering work by James Hardin in the 1970s demonstrated hybridization among 14 of the 16 white oak group species of eastern North America, with hybridization occurring almost anywhere that different white oak species grow in sympatry (Hardin 1975). In the era of DNAbased taxonomy, hybridization has been demonstrated numerous times using chloroplast and nuclear data (Whittemore and Schaal 1991, Dumolin-Lapegue et al. 1997, Curtu et al. 2007, Cavender-Bares and Pahlich 2009). For this reason, oaks have been described by two leaders in the field of speciation as a “worst case scenario for the biological species concept” (Coyne and Orr 2004, p. 43). Our understanding of the depth and orientation of genetic boundaries, our concepts of what constitutes a plant species, and our ability to differentiate morphologically similar species are tangled up in the oaks
A Worst Case in A Genus Of Worst Cases
The Western Great Lakes endemic Hill’s oak (also known as northern pin oak) (Quercus ellipsoidalis; Plate 1) is distinguished by the number of workers who have puzzled over its taxonomic status and proper identification (Trelease 1919; Jensen 1977, 1984; Overlease 1977, 1991; Maycock et al. 1980; Shepard 2009). Hill’s oak is a member of the black oak group, Quercus section Lobatae, a New World lineage of more than 100 species, of which approximately 75 are found in Mexico and 35 in North America north of Mexico. The section is easily recognized in the field by the presence of bristles or awns on the tips of the lobes (in, for example, Q. velutina, Q. rubra, Q. shumardii, Q. palustris) or leaf apex if the leaf is unlobed (for example, Q. imbricaria, Q. phellos, Q. pumila). Most species in the group also mature acorns over two seasons.
In habitat, Hill’s oak ranges from dry sandstone bluffs, oak barrens, and sand savannas to seasonally wet sandy soils and dry-mesic forests in clayey soils. The tree is particularly common in woodlands of northeastern Illinois. Typical Hill’s oaks have deeply lobed leaves with more-or-less C-shaped sinuses; leaf undersides that are smooth or at most sparsely pubescent; terminal buds that are silky-pubescent on the distal (upper) third to two-thirds; and acorn caps that are smooth to sparsely pubescent on the inner surface, with scales on the upper surface that have tightly appressed tips. In these characters, Hill’s oak is similar to the more widespread eastern North American scarlet oak (Q. coccinea; Plate 2), and in fact it was commonly identified as scarlet oak when first viewed by botanists in the late nineteenth century.


In 1891, Reverend Ernest J. Hill encountered a few populations in the area around Glenwood and Calumet Park, Cook County, Illinois that he identified as scarlet oak “with some misgivings.” With further study, Hill judged that the leaf coloration in fall, bark texture, and acorn shape sufficiently distinguished the tree from scarlet oak to warrant its recognition as a separate species, and he published his description of the species in the Botanical Gazette in 1899. Subsequent to this work, many botanists accepted that Hill’s oak was found throughout the upper Midwest to the exclusion of scarlet oak. However, the distinction between Hill’s oak and scarlet oak is not always clear. At their morphological extremes, scarlet oak and Hill’s oak are readily distinguishable. Typical scarlet oak has larger leaves and terminal buds; acorn cap scales with broad, glossy bodies and tips tending to be narrow and somewhat elongate/ acuminate; and concentric rings of pits around the exposed (stylar) end of the acorn nut that appear as though they were scratched with an etching needle or burned into the acorn. Hill’s oak has smaller leaves and terminal buds; acorn cap scales with dull or pubescent bodies and relatively short apices; and usually no rings around the tip of the acorn cap, occasionally one or two small rings. But these characters overlap in the greater Chicago region, especially northwestern Indiana, and as a consequence the taxonomy of these two species has remained in flux.
We began a study at The Morton Arboretum in 2005 to investigate whether Hill’s oak, scarlet oak, and the widespread black oak (Quercus velutina; Plate 3) are genetically distinct from one another. My primary collaborator in this project, Jaime Weber, and I have sampled oaks from 58 sites (Figure 1) and genotyped nearly 700 Hill’s and black oaks as well as populations of scarlet oak from Missouri, southern Illinois, southern Ohio, and upstate New York, and of the related species red oak (Q. rubra), Shumard’s oak (Q. shumardii), and pin oak (Q. palustris).



We are currently investigating three basic questions. First, are Hill’s oak and scarlet oak genetically distinct from one another? Do they show the genetic separation we expect of distinct species? Can we use genetic data to identify morphologically problematic populations in northwestern Indiana and southern Michigan that confound our efforts to understand the natural distribution of Hill’s oak and scarlet oak? Second, do local populations of Hill’s oak and black oak exhibit gene flow, and does genetic intermediacy between these species correlate with morphological intermediacy? Finally, what is the evolutionary history of black oak section members, and what can this history tell us about the process of oak diversification?



Hill’s Oak and Scarlet Oak: Two Different Gene Pools
We began our work uncertain as to whether Hill’s oak and a genetically distinct scarlet oak were both present in the Chicago region. We also did not know whether we would be able to distinguish closely related species at all using genetic data. Previous workers in the region had found that microsatellite data, which is generated by surveying the genome for rapidly evolving repetitive DNA regions, is not consistently able to distinguish such species as white oak and its relatives (Craft and Ashley 2006) or members of the black oak group (Aldrich et al. 2003). We decided to utilize the amplified fragment length polymorphism (AFLP) technique to genotype trees in this study. The AFLP approach is a shotgun-type approach used for DNA fingerprinting and genome scanning. The method entails cutting the genome of an organism into a large number of pieces at arbitrary points in the genome, then using the size distribution of the DNA fragments to estimate genetic similarity between organisms. The disadvantages of AFLP data relative to microsatellite and DNA sequence data is that without directly sequencing AFLP markers, one generally has to assume that markers of a given length are identical by descent and that each marker represents a gene region independent of all others sampled, in which we can identify alleles that are present but not alleles that are absent. These facts render the data less useful for population genetic studies than microsatellite data, but the ability to sample large numbers of genes across the entire oak genome is desirable if we are to detect genetic differentiation even in the presence of interspecific gene flow.
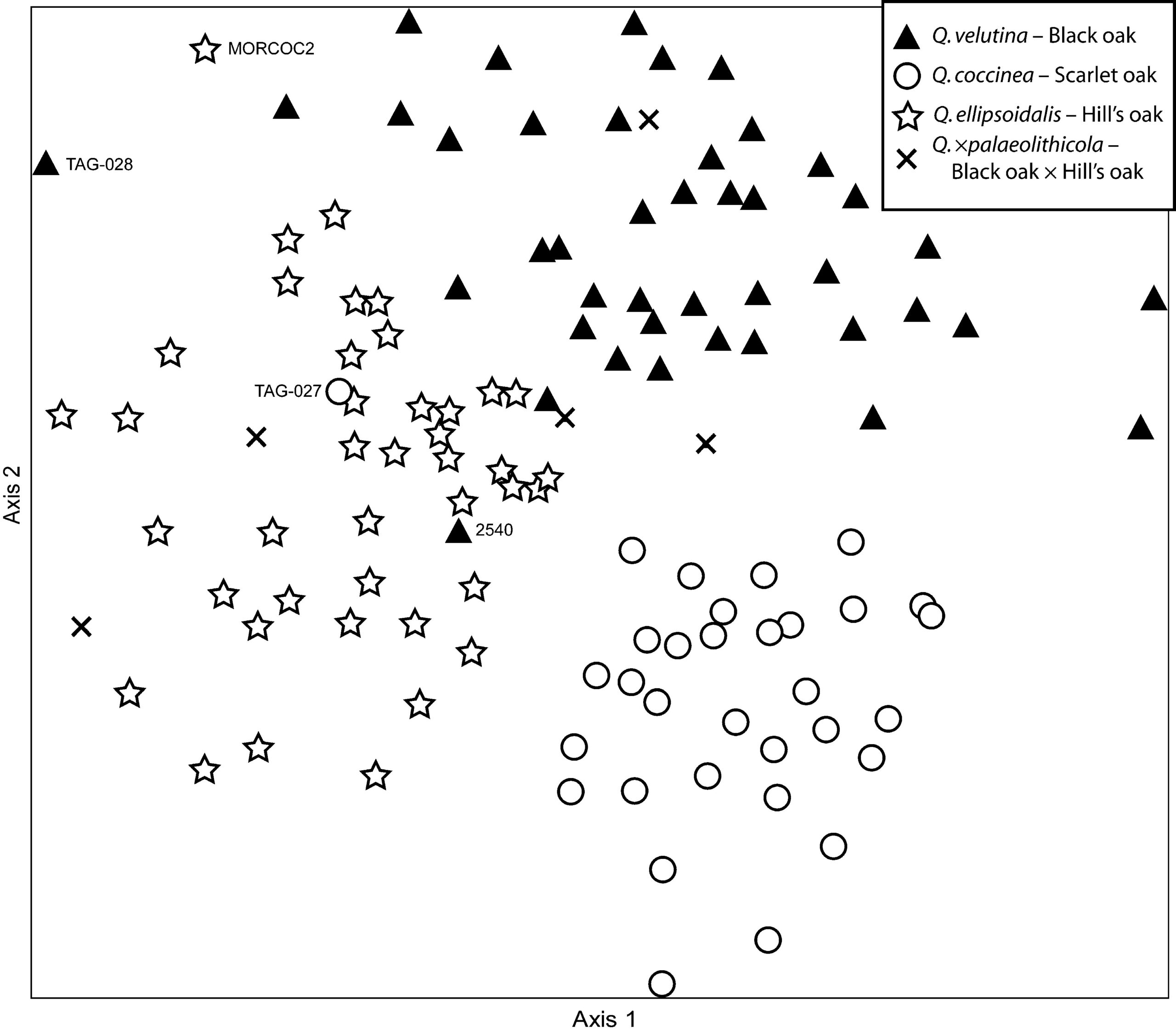

All analyses we have conducted demonstrate a strong separation of scarlet oak from the other species investigated, stronger than the separation between Hill’s oak and black oak (Figure 2). It is important to note that genetic divergence alone does not make a species. It has long been recognized that there can be strong genetic differentiation among populations within species (Ehrlich and Raven 1969). However, when genetic divergence between two putative species exceeds genetic differentiation between other closely related taxa recognized as being distinct at the species level, and when this differentiation is associated with geographic distance (allopatry; Figure 1), most biologists are inclined to recognize the taxa as distinct species. The divergence between scarlet oak and Hill’s oak must be explained either as divergence between two species or as genetic divergence within a single, wide-ranging species. Although geographic distance may play a role in the strong separation between these two species, we have found in follow-up analyses (Hipp and Weber 2008; Hipp et al. unpubl.) that there is little association between genetic differentiation and geographic distance in black oak across a similar geographic range. When we sample Hill’s oaks of northwestern Indiana and southern Michigan that are morphogically similar to scarlet oak (e.g. Figure 2, individual TAG-027), for the most part they do not appear to be genetically similar to scarlet oak, though the genotypes of a small number of samples we have collected in northwest Indiana suggest that scarlet oak may be present in that area. It is significant that we find very few individuals with genotypes intermediate between Hill’s oak and scarlet oak. Naturally-occurring scarlet oak also appears to be rare in the range of Hill’s oak, with a few exceptions. First, as indicated above, our data suggest that scarlet oak may be present in northwest Indiana, based on a few specimens that are genetically intermediate between Hill’s oak and scarlet oak. However, the one specimen we sampled from northwest Indiana that appears morphologically to be unambiguous scarlet oak (TAG-027) genotypes as pure Hill’s oak, and results at other sites where scarlet oak is not present (e.g., central Wisconsin) suggest that occasional genetic assignment discrepancies between Hill’s oak and scarlet oak may be a consequence of genetic similarity between the two species. Our findings on this bear more detailed follow-up work. Second, we have genotyped a few trees from a stand of scarlet oaks and other southern Illinois trees previously reported from Tinley Creek Forest Preserve, Cook Co., IL (Shepard 2005). Scarlet oaks from this site are the only trees in our study to genotype as pure scarlet oak in the Great Lakes region, with no evidence of introgression from Hill’s oak or black oak. However, they appear to have been planted in the twentieth century, as they occur on former oldfield habitat (pers. obs.). Moreover, smaller trees from an adjacent forest margin genotype as scarlet oak as well, though with minimal evidence of introgression from Hill’s oak, and may be natural offspring of these introduced trees. These facts notwithstanding, the strong genetic disjuncture we see between Hill’s oak and scarlet oak gives us a great deal of confidence that the morphological intermediacy between them (Shepard 2009) has more to do with intraspecific morphological variation than with gene flow between them. Hill’s oak and scarlet oak are distinct species.


Black Oak and Hill’s Oak: Gene Flow, But Not as Much as You Might Think
Having determined that Hill’s oak and scarlet oak are genetically distinct from one another, we were interested in understanding the source of genetic similarity between black oak and Hill’s oak. In northern Illinois, Wisconsin, and Michigan, distinguishing these two species from each other is not always straightforward. As is the case with Hill’s oak and scarlet oak, specimens that lie at morphological extremes are easy to identify: typical black oak has large, densely pubescent terminal buds; acorn caps with loose scales and dense, matted pubescence on the inner surface; and leaves that are often pubescent, even roughly so, tending to be less deeply lobed than those of Hill’s oak. However, morphological intermediates are not uncommon (though with good material they are less common than people may suspect), and our first thought was that morphological intermediacy might be predicted well by genetic intermediacy. Our attempt to place morphologically intermediate individuals on our ordinations suggests something different: specimens with mature winter buds and/or acorns as well as reasonably intact leaves and that nonetheless have characteristics of both Hill’s oak and black oak genotype across a wide range of the two species rather than in a position intermediate between them (Figure 2). Other researchers have found similar discrepancy between morphological and molecular estimates of admixture (e.g., Craft et al. 2002, González-Rodríguez et al. 2004), which may be a product of the complex history of crosses and back-crosses expected in a group of outcrossing, readily hybridizing species like the oaks.




Subsequent analysis of our full set of sampled individuals demonstrates a few misclassifications between black oak and Hill’s oak, i.e., incongruence between our identifications based on morphology and the population assignments based on genetic data: 14 black oak out of 286 sampled have > 0.20 assignment to Hill’s oak in a commonly used Bayesian population genetic analysis approach. This mismatch between genetic and morphological species assignments is a hallmark of introgressive hybridization and has been reported previously in oaks (CavenderBares and Pahlich 2009), and the presence of such individuals supports the hypothesis of gene flow between the two species. It is remarkable, however, that we find so little genuine misclassification or evidence of genetic admixture between black oak and Hill’s oak. Our findings build on those of a now-classic study of European oaks (Muir et al. 2000) in demonstrating that while oaks do hybridize, there are enough barriers to interspecific gene flow to make oak taxonomy a meaningful enterprise.
Phylogeny of The Black Oaks: A Little Information, A Lot To Learn
Our work going forward is aimed at understanding how these species and their relatives are related, and how contemporary gene flow and evolutionary history interact to define the limits of today’s oak species. Utilizing a larger AFLP dataset and species sampling, we have found that Hill’s oak and scarlet oak are sister species, meaning that they share a more recent common ancestor than either shares with black oak, red oak, pin oak, or any other species. The morphological overlap we see between Hill’s oak and scarlet oak suggests that the two species may have inherited a similar pool of characteristics from a recent common ancestor, though these characteristics were inherited in differing proportions.
Citation: Hipp, A. L. 2010. Hill’s oak: The taxonomy and dynamics of a western great lakes endemic. Arnoldia, 67(4): 2–14.
This finding is particularly interesting in light of the distribution of Hill’s oak and scarlet oak. Hill’s oak is the only oak species endemic to the Great Lakes region (Abrams 1992) and is distributed almost exclusively in glaciated terrain. It is tolerant of disturbance and has been characterized as the most drought-tolerant of the black oak species (Colodonato 1993), though it appears to be less common than black oak in the driest sand soils of northern Illinois. Its geographic range also overlaps closely with the distribution of dry soil oak savannas in the Great Lakes region (Will-Wolf and Stearns 1999). Scarlet oak, on the other hand, is distributed predominantly south of the edge of the ice sheet at the last glacial maximum. While also tolerant of disturbance and favoring dry sandy or gravelly soils, scarlet oak is not uncommon in mature forests in more mesic soils (Carey 1992). Given the broad geographic extent of scarlet oak and the compressed distribution of hardwood forests during the glacial maximum (Delcourt and Delcourt 1984), these two species likely co-occurred for at least a portion of the Pleistocene. Why, then, has Hill’s oak migrated into postglacial environments while scarlet oak is largely confined to unglaciated terrain? It may be that differences in cold tolerance between the two species govern their relative distributions. Hill’s oak may also be more tolerant of disturbance or of higher pH or finer soil texture. If so, it may have been more able to take advantage of newly opened territory as the vegetation of the savanna regions around the Great Lakes shuffled around rapidly following glacial retreat. This capacity to respond to relatively rapid environmental change may bode well for Hill’s oak in the future. In the shorter term, our growing understanding of oak evolutionary relationships and ecology should allow us to address basic questions about oak distribution and speciation, and guide predictions about how tree species will respond to future climatic and environmental changes.

Acknowledgements
This work has been funded predominantly by The Morton Arboretum, where oaks have been a priority in research and the living collections for decades. Additional funding was provided by grants from The American Philosophical Society and The Hanes Fund of the Michigan Botanical Club. Rachel Davis, Marlin Bowles, and Paul Manos generously provided feedback on a draft of this article.
References
Abrams, M.D. 1992. Fire and the development of oak forests. BioScience 42: 346–353.
Aldrich, P.R., G.R. Parker, C.H. Michler, and J. RomeroSeverson. 2003. Whole-tree silvic identifications and the microsatellite genetic structure of a red oak species complex in an Indiana old-growth forest. Canadian Journal of Forest Research 33: 2228–2237.
Anderson, R.C., and M.L. Bowles. 1999. Deep-soil savannas and barrens of the Midwestern United States. In: R. C. Anderson, J. S. Fralish and J. M. Baskin, eds. Savannas, barrens, and rock outcrop plant communities of North America, pp. 155–170. Cambridge University Press, Cambridge.
Biagi, A. and R.J. Jensen. 1995. The genus Quercus (Fagaceae) in Indiana: Phytogeography and a key to the species. Indiana Academy of Science 104: 11–24.
Carey, J.H. 1992. Quercus coccinea. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Available: https://www.fs.fed.us/ database/feis/plants/tree/quecoc/all.html [Accessed 5 January 2010].
Cavender-Bares, J. and A. Pahlich. 2009. Molecular, morphological, and ecological niche differentiation of sympatric sister oak species, Quercus virginiana and Q. geminata (Fagaceae). American Journal of Botany 96: 1690–1702.
Coladonato, M. 1993. Quercus ellipsoidalis. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Available: https://www. fs.fed.us/database/feis/plants/tree/queell/ all.html [accessed 5 Jan 2010].
Craft, K.J., M.V. Ashley, and W.D. Koenig. 2002. Limited hybridization between Quercus lobata and Hill’s Oak 13 Quercus douglasii (Fagaceae) in a mixed stand in central coastal California. American Journal of Botany 89: 1792–1798.
Craft, K.J. and M.V. Ashley. 2006. Population differentiation among three species of white oak in northeastern Illinois. Canadian Journal of Forest Research 26: 206–215.
Curtu, A., O. Gailing, and R. Finkeldey. 2007. Evidence for hybridization and introgression within a species-rich oak (Quercus spp.) community. BMC Evolutionary Biology 7: 218.
Delcourt, H. R., and P. A. Delcourt. 1984. Ice Age haven for hardwoods. Natural History 93: 22–28.
Dumolin-Lapegue, S., et al. 1997. Phylogeographic structure of white oaks throughout the European continent. Genetics 146: 1475–1487.
Ehrlich, P.R. and P.H. Raven. 1969. Differentiation of populations: Gene flow seems to be less important in speciation than the neo-Darwinians thought. Science 165: 1228–1232.
González-Rodríguez, A., D.M. Arias, S. Valencia, and K. Oyama. 2004. Morphological and RAPD analysis of hybridization between Quercus affinis and Q. laurina (Fagaceae), two Mexican red oaks. American Journal of Botany 91: 401–409.
Hardin, J.W. 1975. Hybridization and introgression in Quercus alba. Journal of the Arnold Arboretum 56: 336–363.
Hill, E. J. 1899. A new biennial fruited oak. Botanical Gazette 27: 204–208.
Hipp, A.L. and J.A. Weber. 2008. Taxonomy of Hill’s oak (Quercus ellipsoidalis: Fagaceae): Evidence from AFLP Data. Systematic Botany 33: 148–158.
Hipp, A.L., J.Weber, and A. Srivastava. In press (2010). Who am I this time? The affinities and misbehaviors of Hill’s oak (Quercus ellipsoidalis). International Oak Journal.
Jaccard, P. 1908. Nouvelles recherches sur la distribution florale. Bulletin de la Societe Vaudoise des Sciences Naturelles 44: 223–270.
Jensen, R.J. 1977. A preliminary numerical analysis of the red oak complex in Michigan and Wisconsin USA. Taxon 26: 399–407.
Jensen, R.J., R. Depiero, and B. K. Smith. 1984. Vegetative characters, population variation, and the hybrid origin of Quercus ellipsoidalis. American Midland Naturalist 111: 364–370.
Johnson, W.C. and C.S. Adkisson. 1986. Airlifting the oaks. Natural History 10/86: 41–46.
Keator, G. and S. Bazell. 1998. The life of an oak: An intimate portrait. Heyday Books, Berkeley.
Kline, V.M. 1997. Orchards of oak and a sea of grass. In: S. Packard and C. F. Mutel, eds. The Tallgrass restoration handbook for prairies, savannas, and woodlands, pp. 3–21. Island Press, Washington, D.C.
Logan, W. B. 2005. Oak: Frame of civilization. W.W. Norton & Company, Inc., New York.
Maycock, P. F., D. R. Daniel, R. Gregory, and A. A. Reznicek. 1980. Hill’s oak (Quercus ellipsoidalis) in Canada. Canadian Field-Naturalist 94: 277–285.
Muir, G., C.C. Fleming, and C. Schlötterer. 2000. Species status of hybridizing oaks. Nature 405: 1016.
Overlease, W. R. 1977. A study of the relationship between Scarlet oak (Quercus coccinea Muenchh.) and Hill oak (Quercus ellipsoidalis E.J. Hill) in Michigan and nearby states. Journal of the Pennsylvania Academy of Science 51: 47–50.
Overlease, W.R. 1991. Genetic relationships between three species of oaks as determined by common garden studies with populations from Michigan, Indiana and Wisconsin. Journal of the Pennsylvania Academy of Science 65: 71–74.
Price, J. 1999. Flight maps: Adventures with nature in modern America. Basic Books, Perseus Books Group, New York.
Shepard, D.A. 2005. The land that time forgot: Southern flatwood oaks and associates of the Tinley Creek Forest Preserve of Cook County, Illinois. International Oak Journal 16: 47–60.
Shepard, D.A. 2009. A review of the taxonomic status of Quercus ellipsoidalis and Quercus coccinea in the Eastern United States. International Oak Journal 20: 65–84.
Trelease, W. 1919. The jack oak (Quercus ellipsoidalis). Transactions of the Illinois State Academy of Science 12: 108–118.
Webb, T. 1981. The past 11,000 years of vegetational change in eastern North America. BioScience 31: 501–506.
Whittemore, A.T. and B.A. Schaal. 1991. Interspecific gene flow in sympatric oaks. Proceedings of the National Academy of Sciences of the United States of America 88: 2540–2544.
Will-Wolf, S. and F. Stearns. 1999. Dry soil savanna in the Great Lakes region. In: R. C. Anderson, J. S. Fralish and J. M. Baskin, eds. Savannas, barrens, and rock outcrop plant communities of North America, pp. 135–154. Cambridge University Press, Cambridge.
Andrew L. Hipp is Plant Systematist and Herbarium Curator at The Morton Arboretum in Lisle, Illinois. Rachel Davis is an artist in Downers Grove, Illinois.
